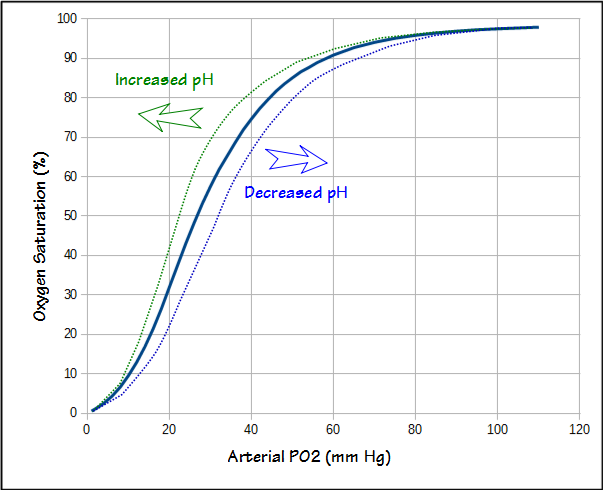
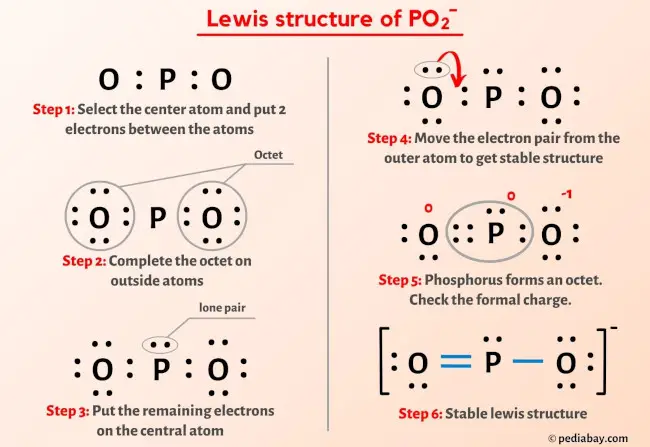
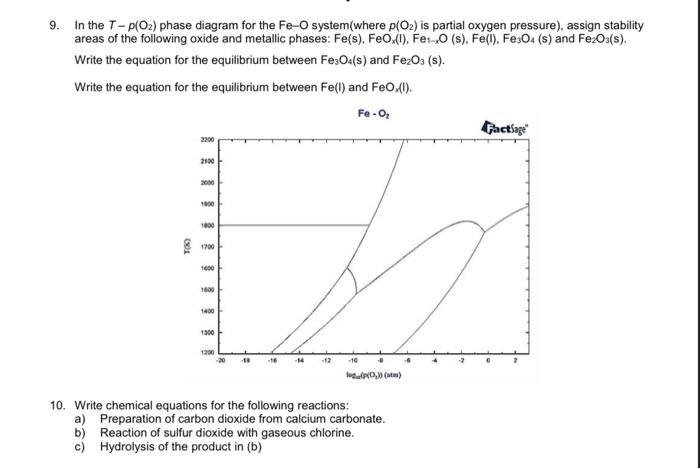
Δμ GS O values at different P O2 , P H2O , and P H2 conditions at 850 °C. | Download Scientific Diagram
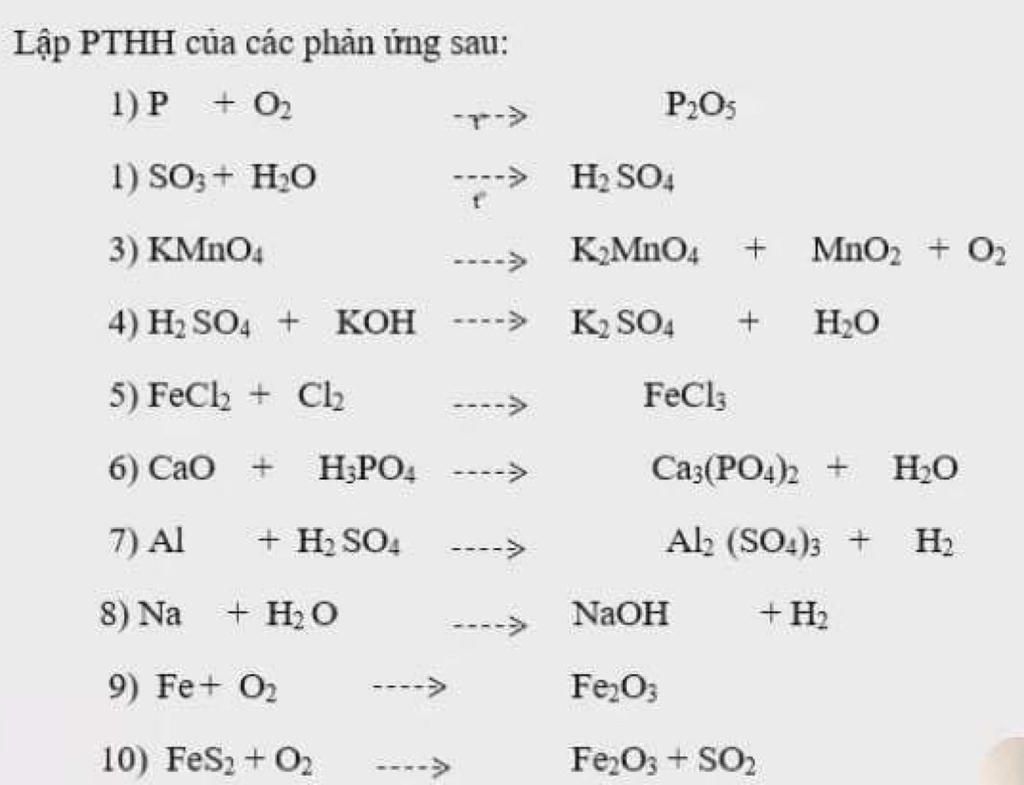
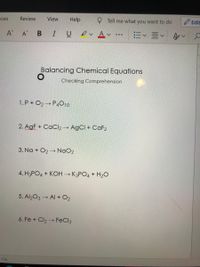
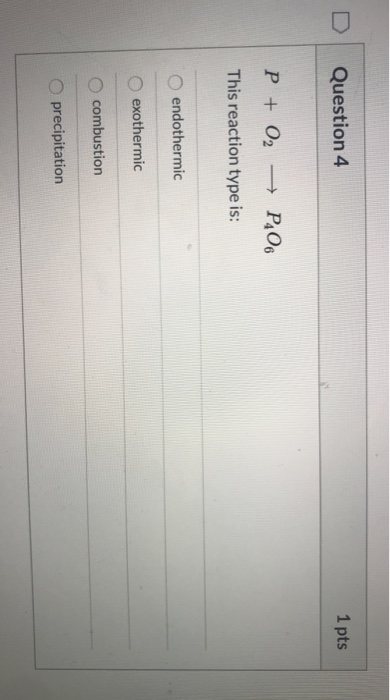
Lập PTHH của các phản ứng sau: 1) P + O2 P2O5 1) SO3 + H2O ----> H2 SO4 3) KMNO4 ----> KMNO4 MnO2 + O2 4) H2 SO4 + KOH ----> K2 SO4 H2O 5) FeClh + Ch --> F