A two litre flask contains 22 g of carbon dioxide and 1 g of helium at 20^0 C. Calculate the partial pressure exerted by CO2 and He if the total pressure is 3 atm.
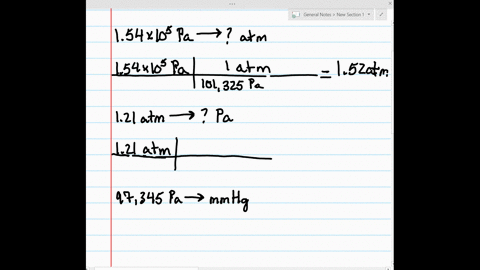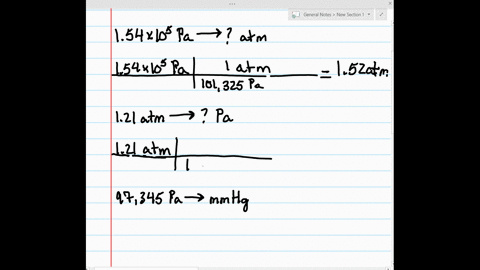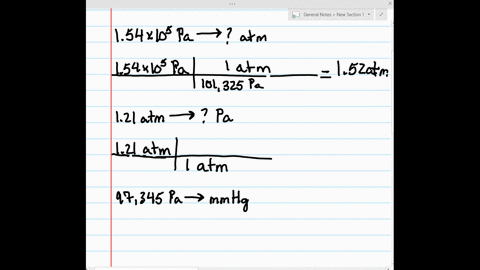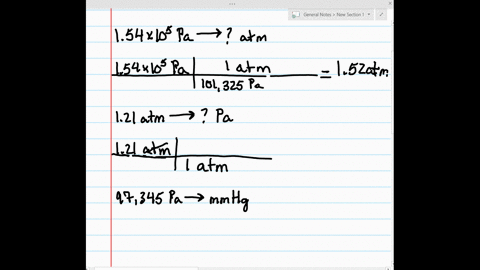
SOLVED:Make the indicated pressure conversions. a. 1.54 ×10^5 Pa to atmospheres b. 1.21 atm to pascals c. 97,345 Pa to mm Hg d. 1.32 kPa to pascals

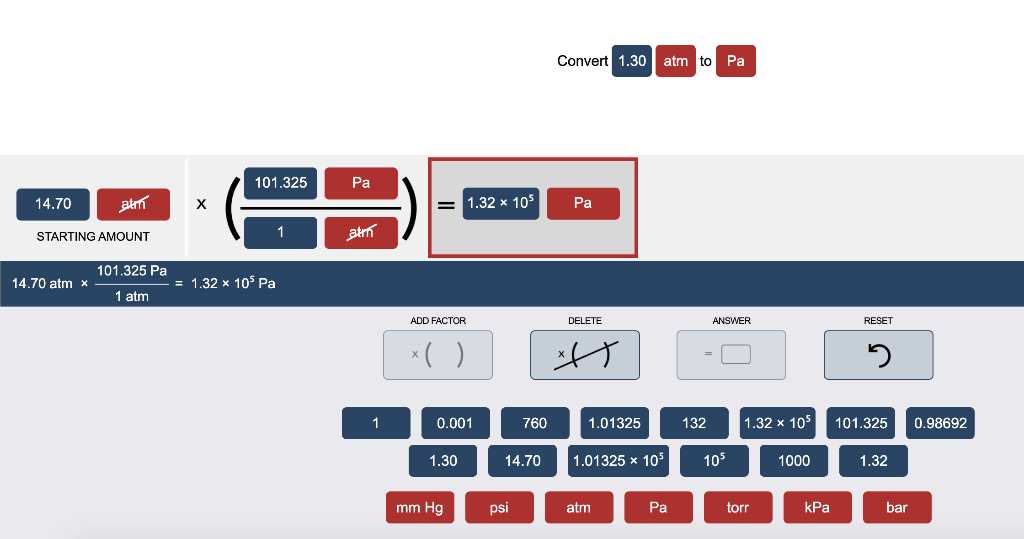
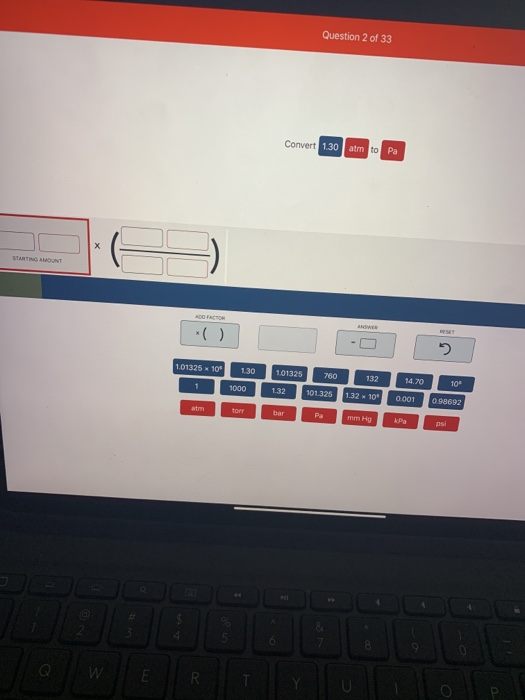
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download

SOLVED:Make the indicated pressure conversions. a. 1.54 ×10^5 Pa to atmospheres b. 1.21 atm to pascals c. 97,345 Pa to mm Hg d. 1.32 kPa to pascals





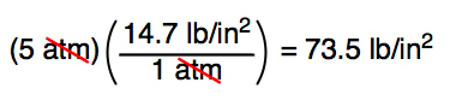





:max_bytes(150000):strip_icc()/sun-beams-diffuse-ocean-micronesia-palau-128941208-587b91ad5f9b584db36312c9.jpg)